 |
| John Dalton 1766-1844 |
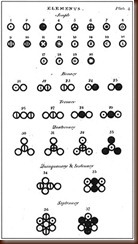 |
| Dalton's atomic and molecular symbolism, from A New System of Chemical Philosophy |
John Dalton also put much thought into color blindness, a condition that he suffered from. He gave a lecture at the Manchester Literary and Philosophical Society, of which he was a member, in 1794, describing the inconsistencies that he observed between how he saw color and how those around him saw colors. He wrote to a friend that “the flowers of most of the Cranesbills appear to me in the day almost exactly sky blue, whilst others call them deep pink.” (The Worthies of Cumberland: John Dalton, p. 101) He also noted that his brother and he agreed on the colors of things, which to modern ears suggests that it was genetic color blindness. Dalton suggested that the cause of the difference between his vision and others was that the fluid in his eye was tinted blue. As a true scientist, he suggested that his eyes should be dissected after his death to see if this was true. It was not, but the eyes were preserved by the Manchester Literary and Philosophical Society and recently the DNA was examined, showing that Dalton lacked one of the three photopigments in the eye. (If you wish to see the present state of his eyes and related images, I suggest you go to http://www.sciencephoto.com/set/803.) This theory of photopigments had been proposed by Thomas Young (1773-1829), one of Dalton’s contemporaries who established the wave theory of light, but even though Young’s view was more correct, color blindness has been historically called Daltonism. This just goes to show that you don’t have to be right to be remembered, you just have to be the first, or perhaps the clearest.
Selected Works by Dalton
- A New System of Chemical Philosophy, vol. 1. (Manchester, Russell), 1808.
- Meteorological Observations and Essays, 2nd edition. (Manchester, Harrison and Crosfield) 1834.
- “Extraordinary Facts Relating to the Vision of Colours: with Observations”. Memoirs of the Literary and Philosophical Society of Manchester 5 (1831) 28-45.
- Kristine Krug, "Science celebrates 'father of nanotech'", BBC News, October 10, 2003.
- Robert Angus Smith, Memoir of John Dalton and History of the Atomic Theory up to his Time. (Manchesterr, Sowler and Sons), 1856.
- Henry Roscoe and Arthur Harden, A New View of the Origins of Dalton’s Atomic Theory: A Contribution to Chemical History. (London, Macmillan and Co.), 1896.
- John Millington, John Dalton. (London, Dent & Co.), 1906.
- D. M. Hunt, K. S. Dulai, J. K. Bowmaker, and J. D. Mollon, “The Chemistry of John Dalton's Color Blindness”. Science 17 (February 1995) 984-988. DOI: 10.1126/science.7863342.
- Henry Lonsdale, The Worthies of Cumberland: John Dalton. (London, Routledge and Sons), 1874. This includes a selection of letters to and from Dalton about colorblindness.
- Thomas Young, “Bakerian Lecture: On the Theory of Light and Colours”. Philosophical Transactions of the Royal Society of London 92 (November 12, 1801) 12-48. DOI: 10.1098/rstl.1802.0004.
Dalton, yay! I definitely remember him from high school chemistry class...and in fact honored him with a song at one point (which garnered some extra credit). Our textbook neglected to mention his offering up his eyeballs to science. A true scientist, indeed!
ReplyDeleteI would like to see more chemists on your blog. Though he isn't quite as significant as the figures you've covered, I would nominate Borodin out of pure curiosity.
By the way, my nomination of Borodin was partly inspired by his inclusion in A Master of Science History: Essays in Honor of Charles Coulston Gillispie, the latest issue of the journal Archimedes. Never had heard of this issue, of Gillispie, or of Archimedes before today. Are you acquainted with this journal? It's pretty interesting! http://www.springer.com/series/5644
ReplyDeletehttp://www.springerlink.com/content/978-94-007-2626-0/